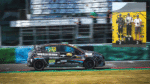This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Man had ‘active role’ moving and storing €280,000 in drugs
A man who had an “active role” in moving and storing almost €280,000 of drugs has been jailed for three and a half years, reports ... Read More

Tallaght ‘moderately littered’ according to IBAL report
The latest IBAL report describes Tallaght as “moderately littered” and the controversial littering site between Lidl and Priory Market was brought up once more. Set ... Read More
Man drove at speed mounting onto footpath missing buggy
A man narrowly avoided hitting a double buggy at a bus stop after causing €11,200 worth of damage to a garda car, reports Eimear Dodd. ... Read More
The changing landscape of Clonburris
Clonburris is a prime example of the ever-changing landscape that exists in Dublin South West, as it has undergone an extreme transformation in the past ... Read More

Calls to finally repair boom at leisure centre swimming pool
Calls have been made by members of the SDCC to finally repair the boom in the Clondalkin pool. Clondalkin Leisure Centre has had continuous issues ... Read More
67 Laurels charity crew cyclists raise money for CHI
The Laurels Cycle Crew accomplished a huge achievement this year with a roundtrip cycle from Clondalkin to Offaly. The team of 67 cyclists pedaled to ... Read More
New local Community Partnerships marks significant step forward
Five new Local Community Safety Partnerships (LCSPs) have been established by Dublin City Council across the five administrative areas. The Local Community Safety Partnerships will ... Read More

Play follows a group of adults with disabilities
Smashing Barriers, a theatre group set up by Disability Action Collective, held a performance of an original play, “The House on the Hill” in the ... Read More
Alex claims fastest lap award at Grand Prix
Alex Hendrick of Drimnagh Castle Secondary School claimed the fastest lap award at the inaugural Kinia Hydrogen Grand Prix (H2GP) test track' during the LogisTYcs ... Read More
Students crowned Money Smart Challenge winners
A team from Adamstown Community College in Lucan have been crowned champions of the 2026 Bank of Ireland Money Smarts Challenge. Close to 2,000 students ... Read More

Bus lanes used as ‘informal parking’
Informal parking arrangements along the Adamstown Link Road “is not giving rise to significant or immediate road safety concerns”, according to the SDCC. The issue ... Read More
Drama group grant for summer school
Lucan Drama Group has found itself in the spotlight following the allocation of a grant totaling €2,975. The group applied for the grant from the ... Read More
Citywest logistics facility on the market at guide price €25.6 million
A 98,000 sq ft logistics facility in Citywest Business Campus set to be delivered by Sandymark Group has been put on the market with a ... Read More
Barbers and coffee shop to replace flower shop
A new barbers and coffee shop is set to be introduced at Springfield Shopping Centre after a change of use was granted to replace a ... Read More
Proposals to demolish stone cottages refused
PLANNING has been refused for the demolition of existing stone cottages on which works have commenced at Hayden House and cottages on Main Street Rathcoole. ... Read More